Unlocking Cleaner Fuels: Revolutionizing Catalyst Stability for Syngas Production
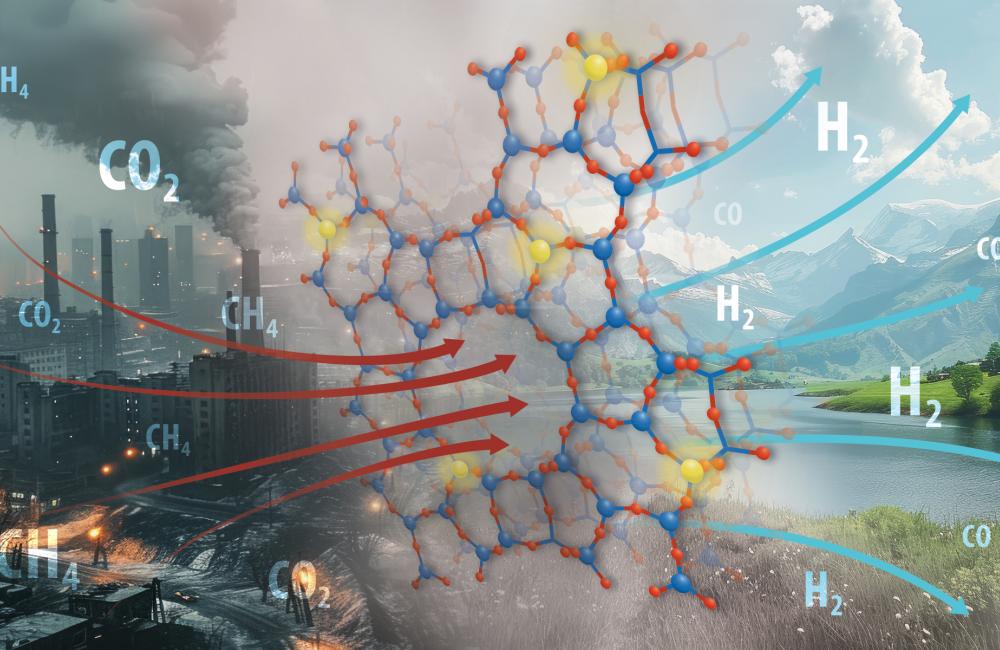
A breakthrough by the Department of Energy's **Oak Ridge National Laboratory** has improved catalyst stability for the *dry reforming of methane*, a chemical process converting harmful **greenhouse gases**—methane and carbon dioxide—into valuable **syngas**. Syngas, a mixture of hydrogen and carbon monoxide, is crucial for producing cleaner fuels and essential chemicals like methanol and ammonia. Despite its benefits, the process is plagued by catalyst deactivation at high temperatures due to coke formation and sintering. The ORNL team tackled these issues by synthesizing a **zeolite-based catalyst** with nickel atoms, creating a framework that stabilizes the metal active sites and prevents these deactivation processes. With zeolite’s intricate, pore-filled structure, the catalyst enjoys a high surface area for reactions while being resistant to degradation at elevated temperatures. **Infrared spectroscopy** and **X-ray absorption studies** confirmed the catalyst's structure and efficiency, which was achieved through a synthesis method that eliminates destabilizing water. This innovation not only holds the potential to improve global **energy security** but also proposes a *sustainably innovative* design principle for a broad range of industrial catalytic processes. The implications extend beyond this single catalyst, suggesting pathways to design new, robust catalysts through **rational design** rather than trial and error, marking a significant step forward for industrial sustainability and environmental responsibility.